 See how to use this calculator for step-by-step guide. Introduction to Chemical Engineering Thermodynamics (8th ed.). The entry required is in Fahrenheit degrees there is a converter below this calculator if you only know the Celsius temperature for the entries required. Bear in mind that flash calculation can't be calculated ifįeed exists as subcooled liquid or superheated vapor. 
Where \(z_i\) is molar composition of the inlet feed, \(V\) is the vapor fractions, and \(L\) is the liquid fractions or \(L=1-V\). While for dew point calculations, the unknown liquid-phase compositions can be simplified by asumming \(\sum x_i=1\), so thatįor flash calculation, we calculate the composition based on material balance of the system, Use our dew point calculator to calculate either pressure dewpoint to atmospheric dewpoint or atmospheric dewpoint to pressure dewpoint. P_i^, given \(P\) and \(T\)įor bubble point calculations which vapor-phase compositions are unknown, we can simplify the calculations by asumming \(\sum y_i=1\), so that the Raoult's law become Although most dew points are stated at atmospheric pressure, pressure dew point should be considered, because condensation can occur in the air pressure lines if the temperature decreases. to provide Uvalues, condensation / dew point risk, required insulation thicknesses etc Uvalue calculations can also be carried out on the. The mathematical equations which reflects the Raoult's law based on the two assumptions above is Fase cair adalah larutan ideal, yang berarti hanya berlaku jika spesies kimia yang menyusunnya mirip secara kimiawi.Fase uap adalah gas ideal, yang berarti hanya dapat digunakan dari tekanan rendah hingga sedang, dan.Please provide any two of the three variables below to calculate the third. Ada dua asumsi utama dalam hukum Raoult, yaitu: This calculator estimates the temperature to which air must be cooled to become saturated with water vapor and form dew. Hukum Raoult menyatakan bahwa tekanan uap pelarut di atas larutan sama dengan tekanan uap pelarut murni pada suhu yang sama yang diukur dengan fraksi mol pelarut yang ada. Untuk melakukan perhitungan bubble dan dew point, terlebih dahulu kita harus memahami teori Hukum Raoult untuk campuran ideal. 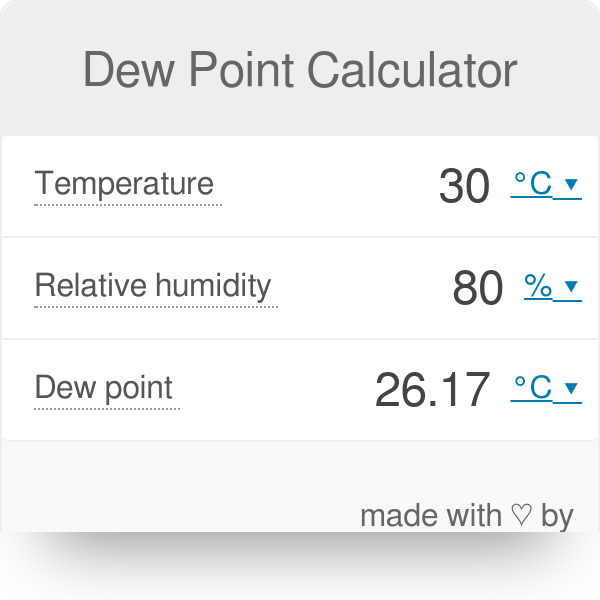 
Titik Gelembung, Titik Embun, dan Perhitungan Kilat 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
